Cannabis laws in Europe FAQ: What laws cover medical and commercial cannabis-derived products?
This page is part of the Cannabis laws in Europe FAQ which answers some of the more frequently asked questions raised in discussions about cannabis legislation.
Last revised: 21 April 2026
Introduction
This part of the Cannabis laws in Europe FAQ provides an overview of legislation around the legal use and supply of cannabis in Europe. The EU has many directives and regulations that may be applicable to low-THC cannabis products in their different forms and components. This includes the medical use of cannabis, and low-THC products, such as wellness products, cosmetics and food, including food supplements. Some of these laws may apply automatically and uniformly to all EU Member States; others will need to be transposed into national law.
Is the medical use of cannabis legal?
As the UN conventions call for the drugs under international control to be limited to ‘medical and scientific purposes’, there is scope under international law to allow cannabis, or cannabis-derived products to be used as a medicine to treat certain defined conditions. This is reflected in the fact that there are many narcotic substances listed in the drug control conventions that have an authorisation for use in or as medicinal products in the European Union.
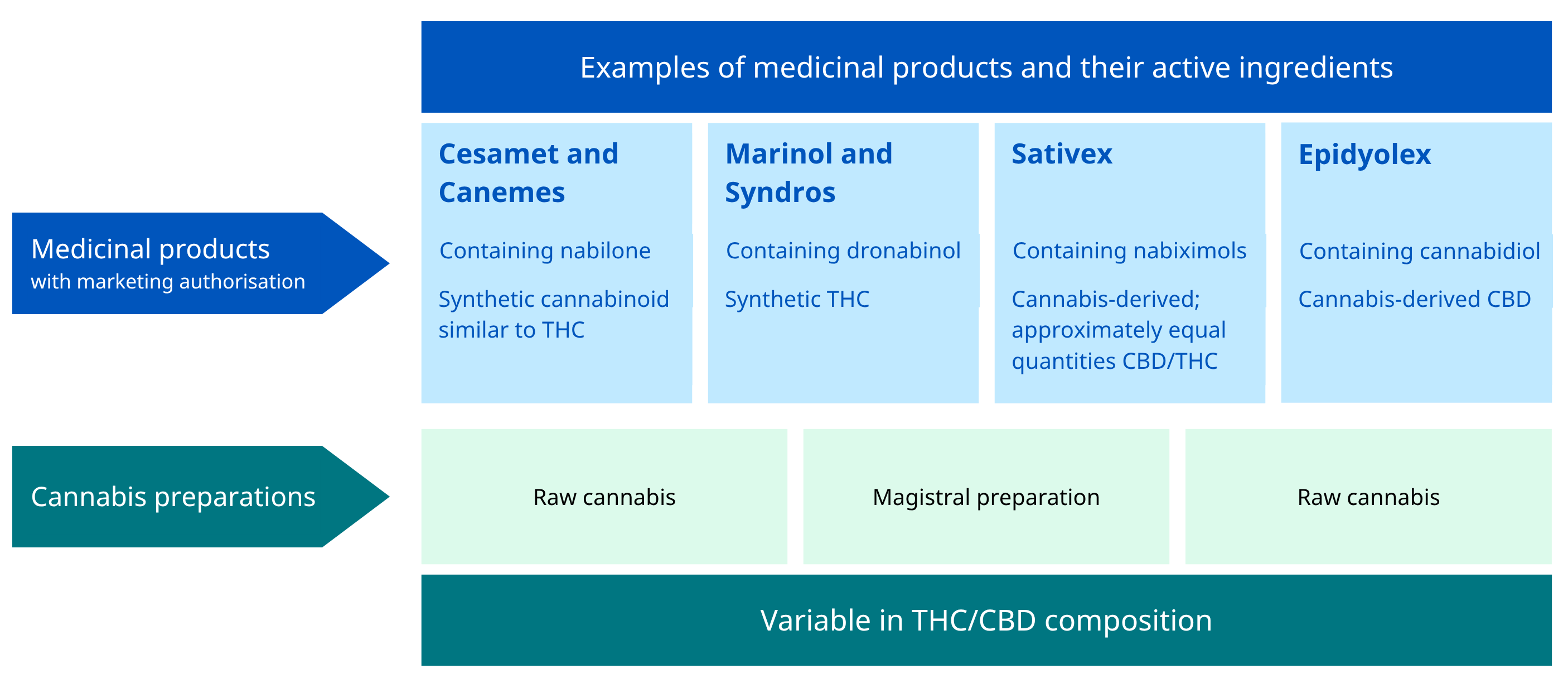
In the public debate, the term ‘medical use of cannabis and cannabinoids’ has been used non-technically and non-consistently to refer to a wide variety of preparations and products (see Figure 8) that may contain different active ingredients and be taken through different routes of administration. Although in practice some of the terms in this area have often been used rather loosely, the distinctions between them can have both regulatory and medical implications.
It is important to note the term ‘cannabis and cannabinoids for therapeutic purposes’ includes medicinal products with marketing authorisation and cannabis preparations being made available through other regulatory measures for therapeutic purposes.
Show a text version of the above graphic
Examples of medicinal products and their active ingredients (Medicinal products with marketing authorisation):
- Cesamet and Canemes
- Containing nabilone
- Synthetic cannabinoid similar to THC
- Marinol and Syndros
- Containing dronabinol
- Synthetic THC
- Sativex
- Containing nabiximols
- Cannabis-derived; approximately equal quantities CBD/THC
- Epidyolex
- Containing cannabidiol
- Cannabis-derived CBD
Examples of cannabis preparations (Variable in THC/CBD composition):
- Raw cannabis
- Magistral preparation
- Standardised cannabis preparations
Having a marketing authorisation means that an application for a medicinal product was submitted to a regulatory authority and, after evaluating the application, the regulatory authority granted authorisation. This usually implies that the product went through extensive clinical trials and that the drug has been tested for safety, efficacy and side effects. Regulatory authorities also consider whether the product can be manufactured to a required quality level.
There are several ways for medicines to receive cross-national marketing authorisation within Europe; and cannabis-derived medicinal products are subject to the general requirements applicable to medicinal products. Further information on approval procedures can be found on the dedicated websites of the European Commission and the European Medicines Agency (EMA, no date).
Cannabis-derived medicinal products can be authorised in the EU after their safety, efficacy and quality are assessed in line with the EU pharmaceutical legislation. As a result, cannabis-derived medicines are already available on the EU market (for example Epidyolex).
In the absence of such authorisation, some Member States may allow patients access to cannabis-derived medicinal preparations, when such a preparation is prescribed to an individual patient by a medical doctor, via an exception provided in the EU pharmaceutical legislation, that is, Article 5 of Directive 2001/83/EC.
The Committee on Herbal Medicinal Products of the EMA has compiled a list of terms and definitions for cannabis-derived medicinal products, as a summary of relevant scientific and legislative terminology (Committee on Herbal Medicinal Products, 2021).
Several countries in Europe permit the cultivation of cannabis for medicinal purposes, with the cultivation and subsequent processing taking place under the usually strict rules that are applicable to agricultural, manufacturing, distribution, security and clinical good practice. Broadly speaking, the cultivated cannabis is intended either for domestic use or for export. The countries with programmes of cultivation primarily destined for domestic use include Czechia, Denmark, Germany, Italy and the Netherlands, with a new law in Poland allowing this from May 2022, while those cultivating cannabis primarily for export include Austria, Greece and Portugal.
In 2018, a summary of the evidence on the effectiveness of cannabis and cannabinoids as medicine found that some cannabinoids can relieve the symptoms of some illnesses, although they are often used as adjunctive treatments (meaning that they are added to other medical therapies) and are typically used after a patient has failed to respond to recommended treatments for these conditions (Hall, 2018) (1). As significant knowledge gaps and considerable uncertainty exist when interpreting the currently available evidence in this area, there is a need for additional research and clinical studies, including larger and better-designed trials. There is also a need for more studies that explore issues of dosage and interactions between medicines, as well as those that include a longer-term follow-up of participants. It is interesting to note in this context that in February 2019 the European Parliament passed a resolution calling for more research into the medical uses of cannabis. There is also currently a lack of research evidence available about the potential abuse and dependence liability of cannabis-derived medical products and what measures might be effective in mitigating any risks in this area (Cooper and Abrams, 2019).
In addition to cannabis-derived medicinal products with a marketing authorisation, a number of EU countries allow patients to use cannabis preparations for medical purposes. The most common initial approach has been to set up some form of special access scheme, typically by creating a system that provides a certain degree of medical approval and oversight, limits use to a restricted set of medical conditions and often restricts the cannabis preparations that patients can use. The decision to subsidise or reimburse patient costs, or for patients to pay full price for the medicine or preparation will also have an impact on the extent of use.
Member States vary in terms of whether cannabis preparations not currently available for prescription can be brought into their territories by non-EU visitors from countries where they are legal with an accompanying medication prescription, and if so in what quantities.
#EMBED-cannabis-laws-faq-box-10#
Are products containing low THC levels legal?
In recent years an increasing number of cannabis products, including herbal cannabis and cannabis oils, have been offered for open sale in Europe. There also appears to be a growing commercial interest in developing products that contain cannabidiol (CBD) or other extracts of the cannabis plant, but without tetrahydrocannabinol (THC), or with only very low levels of THC present. These low levels mean the products might not be controlled under drug laws in some countries, but manufacture and sale may be limited by other trade regulations, and the approach used may not be uniformly applied in different Member States.
A recent EMCDDA study found that low-THC cannabis products are being offered for sale in most EU countries, with a wide variety of retailers active in the market for low-THC products in Europe (EMCDDA, 2020a). While products containing material from the cannabis plant can be found in everyday retail outlets (e.g. health food chain stores, chemists and cafés), there are also dedicated shops selling low-THC cannabis products. Some of these focus on health and well-being, while others appear to be concentrating more on products that look similar to those that exist on the illicit recreational cannabis market. This means that products containing extracts of the cannabis plant are appearing in a number of different commercial sectors where different regulatory frameworks operate. In some cases, this is also creating tension with drug control regulations.
These developments have given rise to concerns at the policy level regarding the legal status of these products and their potential to cause harm. A specific challenge therefore that is faced by both policymakers and those wishing to supply low-THC products lies in establishing the legal status of low-THC products and which regulatory frameworks should apply to their sale. The EU has many directives and regulations that may be applicable to low-THC cannabis products in their different forms and components, and which provide standardised definitions of different types of products, such as foods and cosmetics (see subsequent sections). Some of these may apply automatically and uniformly to all EU Member States; others will need to be transposed into national law.
Other phytocannabinoids (cannabinoids produced in plants), such as CBN (cannabinol), CBC (cannabichromene) and CBG (cannabigerol), have in recent years been the subject of scientific studies investigating their use for medicinal, cosmetic and other purposes. The regulatory frameworks for such products are still developing.
Is it legal to sell low-THC cannabis products for smoking?
Low-THC cannabis products can be sold for smoking in some countries, provided they comply with certain regulations.
Herbal products that are marketed for smoking, claiming to have a low THC content and/or as being extracted from industrial cannabis plants, have been openly offered for sale in many EU countries since 2017. An EU-wide policy instrument is the regulatory system established in EU Directive 2014/40/EU on tobacco and related products (‘Tobacco Products Directive’, TPD). The Directive defines ‘herbal products for smoking’ as ‘a product based on plants, herbs or fruits which contains no tobacco and that can be consumed via a combustion process’. The Directive in itself does not exclude the possibility that cannabis or related products may be regulated as herbal products for smoking. However, if these products are legally placed on the market and deemed to fall within the definition of ‘herbal products for smoking’, they will need to comply with the TPD.
If products are classified as falling within the remit of the TPD, Articles 21 and 22 make specific provisions for product labelling and for the reporting of ingredients. Prior to placing a new herbal product for smoking on the market, manufacturers and importers need to submit to the competent national authorities a list of ingredients and respective quantities by brand name and type. According to the submitted information, herbal products for smoking which contain or may be otherwise associated with cannabis have been reported in most Member States to date, with an increase observed in the number of products since 2019.
The sale of low-THC cannabis herb and resin products can pose a new challenge for law enforcement, as distinguishing between low- and high-THC cannabis on the street, in shops or at the border is not simple, and testing all products would be impractical and costly. In countries such as Austria, Italy and Switzerland, police now have a rapid reagent test for use on the street. Some portable tests can analyse whether a product contains THC or not and others can indicate the amount of THC present. However, not all law enforcement agencies across Europe have these instruments available. It is also important to note that it can be difficult to achieve consistent test results from the same product, even when tested in a laboratory (Giese et al., 2015).
This issue from a policy perspective is further complicated by the reported adulteration of low-THC herbal and resin cannabis with potent synthetic cannabinoids, which are then sold as illicit cannabis on the illicit drug market (EMCDDA, 2022).
Is cannabis legal as a wellness product?
Low-THC cannabis products might be sold as ‘wellness products’ in some countries, provided they comply with certain regulations.
The concept of a wellness product has no formal legal recognition at the EU level, but it is a popular non-scientific term which is loosely used to describe a product aiming to optimise physical, mental or social well-being. As such, wellness products may be included within several different EU regulatory frameworks, depending on their intended use. To answer the question of whether cannabis is legal as a wellness product, we briefly consider the current situation and the legislation that might be applied; more details can be found under the specific sections of this report.
While a large number of different cannabis wellness products have appeared on the European market, the most widespread cannabinoid marketed in these products is CBD. Several countries in Europe have treated CBD as an ‘extract of cannabis’ and thus a controlled drug in accordance with the 1961 UN Convention.
Since the Court of Justice of the European Union (CJEU) decision in November 2020 (see the box below), CBD has been recognised as not falling within the scope of narcotics control laws when interpreting EU laws — though some countries may choose to maintain CBD in their lists of controlled substances.
Broad health claims appear to be a significant contributor to the demand for CBD-related products. However, despite claims that these products may be useful for treating a wide range of illnesses or symptoms, there is currently insufficient evidence available with respect to many conditions to enable an informed assessment of the veracity of these claims. According to Regulation (EU) no 1169/2011 on the provision of food information to consumers, any claims that a product prevents or treat disease, or relieves symptoms, are not permitted, and would bring these products under the scope of medicines regulations, requiring them to have a licence for sale (see Is the medical use of cannabis legal?). To avoid this, the marketing or reviews of these products tend to use non-specific words or phrases, often claiming that CBD improves ‘well-being’ or something similar, but without directly stating that these products have medicinal properties.
Any claims made for foods that they are beneficial for health should be authorised under Regulation (EC) No 1924/2006 on nutrition and health claims made on foods (Claims Regulation), after a scientific assessment of the highest possible standard, for which the European Food Safety Authority (EFSA) is responsible. Also, reference to general, non-specific benefits of the nutrient or food for overall good health or health-related well-being may only be made if accompanied by a specific health claim included in the Union list of permitted health claims.
To date, no such health claims have been authorised for cannabis products, although there are submissions for authorisation on the health claims of hempseed oil (Cannabis sativa) and on hemp-agrimony (Eupatorium cannabinum) (leaves) that are awaiting a final decision by the European Commission (see also the section Is cannabis legal as a food or food supplement?). Pending a final decision, the use of health claims for botanical substances has been put ‘on hold’, although these may continue to be used provided that they comply with the general principles and conditions of the Claims Regulation and the relevant national provisions.
Court of Justice of the European Union ruling on CBD, November 2020
In 2018 a business in France imported low-THC cannabis oil from Czechia and began selling it to the public in e-cigarette cartridges, promoting its CBD content. The oil was made from the whole cannabis plant, which was legal in Czechia but not in France, where the commercial use of hemp was restricted to fibre and seeds. The business owner was convicted, and appealed. The case was referred to the Court of Justice of the European Union (C-663/18), which declared in November 2020 that while the CBD in the case was not a drug within the meaning of the 1961 Convention, the fundamental principle of free movement of goods between Member States could still be limited on grounds of protecting human health and life. However, such a limit should not go beyond what is necessary in order to achieve its objective and should be applied in a consistent and systematic manner. The court stated that, while the evidence for CBD posing a risk to health, although still limited, may justify precautionary restrictive measures, it was inconsistent to apply the marketing ban only to organic, and not synthetic, CBD. The court’s statement that CBD was not a drug within the meaning of the 1961 Convention has implications for interpretations of the EU laws discussed here.
Are cosmetic products containing cannabis legal?
Some parts of the cannabis plant may be used in cosmetics; others are expressly prohibited. The EU Regulation on cosmetic products, Regulation (EU) No 1223/2009, Article 2(1)(a) defines cosmetics as ‘any substance or mixture intended to be placed in contact with the external parts of the human body (epidermis, hair system, nails, lips and external genital organs) or with the teeth and the mucous membranes of the oral cavity with a view exclusively or mainly to cleaning them, perfuming them, changing their appearance, protecting them, keeping them in good condition or correcting body odours’.
Annex II of this Regulation lists substances prohibited in cosmetic products. All substances in Tables I and II of the 1961 Convention are incorporated by entry 306 of this annex, including cannabis and extract of cannabis. However, as described earlier in this report, the 1961 Convention defines cannabis as ‘the flowering or fruiting tops of the cannabis plant’ and excludes the seeds and leaves of the plant when not accompanied by the flowering or fruiting tops (2). CBD is not a prohibited substance in cosmetic products, as it is not covered by Tables I and II of the 1961 Convention in light of the CJEU ruling in case C-663/18 (see the box Court of Justice of the European Union ruling on CBD, November 2020) and consequently, by entry 306 of Annex II.
Several cosmetic ingredients derived from the cannabis plant are listed in the EU Cosmetic Ingredients database (CosIng). As announced on the introductory page to the database, even if an ingredient is assigned an INCI name (International Nomenclature of Cosmetic Ingredients) that appears in the inventory section of CosIng, this does not necessarily mean it is to be used in cosmetic products nor that it is approved for such use. In line with this, some listed ingredients derived from the cannabis plant’s flowers are prohibited for use under the EU Regulation on cosmetic products, although others that are derived from the plant’s roots or seeds, are not. Some listed ingredients are subject to a noted restriction, such as ‘II/306’ (considered a cannabis extract under the 1961 UN Convention) but others, including cannabigerol and cannabidiol, which are used in some skin conditioning products, are not. For some of these listed ingredients, CosIng notes that national legislations on controlled substances may also apply.
Article 3 of the Regulation on cosmetic products imposes a general condition that all such products made available on the market must be safe for human health when used under normal or reasonably foreseeable conditions of use. The same Regulation requires notification of new products on the EU market at the EU Cosmetics Products Notification Portal, and several cosmetic products containing CBD can be found there.
Is cannabis legal as a food or food supplement?
When some parts of the hemp plant or hemp-derived products are placed on the market as ‘food’ in line with the General Food law, numerous other regulations may apply. Some of these are listed below (3).
In Regulation (EC) No 178/2002 laying down the general principles and requirements of food law, Article 2 states that the definition of ‘food’ is any substance or product intended to be or reasonably expected to be ingested by humans, but it does not include medicinal products, cosmetics, or narcotic or psychotropic substances within the meaning of the 1961 and 1971 Conventions, implying that substances regarded in the conventions as narcotic or psychotropic substances cannot be classified as a food. The national authorities of the EU Member States are responsible for determining whether each product qualifies as a medicinal product or as a foodstuff. Producers may also have conflicting interests in the classification of their product; while medicinal products require extensive trials, they attract zero import duty, in contrast to food products, which are subject to considerable duty tariffs.
The Regulation on novel foods ((EU) 2015/2283), which repealed Regulation (EC) No 258/97, defines novel foods as food that had not been consumed to a significant degree by humans in the European Union before 15 May 1997. Novel foods may only be placed on the market within the European Union subject to an authorisation, and only after they have been found safe by EFSA.
Directive 2002/46/EC on the laws relating to food supplements states that food supplements are foodstuffs (i.e. foods) that are concentrated sources of minerals or vitamins or other substances with a nutritional or physiological effect, intended to supplement a normal diet and marketed in dose form.
Cannabis products including extracts, which are not considered narcotics, medicines or cosmetics might be considered a food, provided that all other conditions of Article 2 of Regulation (EC) No 178/2002 are met. Hemp seed and hemp-seed derived products, such as flour, are considered traditional foods. A number of applications for CBD products — including synthetic CBD — have been made, seeking EU approval as novel foods. According to the Regulation on novel foods ((EU) 2015/2283)), food business operators must verify whether or not the food which they intend to place on the EU market qualifies as a novel food, consulting the relevant national authorities as necessary.
The EU Novel food catalogue is a non-exhaustive and non-binding database which presents the outcome of discussions within the European Commission Working Group on Novel Foods with regard to whether or not specific foods were used for human consumption to a significant degree within the European Union before 15 May 1997. The catalogue can indicate the position taken by Member States on certain products, but it does not replace the various legally mandated procedures. The catalogue currently has three entries relevant to this report, Cannabis sativa L., cannabidiol (CBD) and cannabinoids. Under the heading Cannabis sativa, the catalogue observes that some products derived from the Cannabis sativa plant, or plant parts such as seeds, seed oil, and hemp seed flour, have a history of consumption in the European Union and are therefore not novel. However, national legislation may restrict the marketing of some of these foods. Since January 2019, the entry on CBD has also directed the user to the entry ‘Cannabinoids’ which states that extracts of Cannabis sativa and derived products containing cannabinoids, and products containing cannabinoids as added ingredients, as well as synthetically obtained cannabinoids, are considered novel foods as a history of consumption has not been demonstrated.
Food business operators can place a novel food on the EU market only after the European Commission has processed an application for the authorisation of a novel food. This includes verification of whether the specific product falls within the definition of ‘food’ (see definition above) and the performance of the safety evaluation by EFSA, and has adopted an implementing act authorising placing a novel food on the market.
In recent years there has been an increase in applications (Norwinski et al., 2019) for the authorisation of CBD-containing products under the EU Regulation on novel foods. Following the decision of the CJEU in Case C-663/18 (see Is cannabis legal as a wellness product?), the European Commission noted that CBD should not be considered as a narcotic drug within the meaning of the UN Single Convention on Narcotic Drugs of 1961. It also noted that CBD can be qualified as a food, provided that the other conditions of EU food-related regulations, such as the EU Regulation on novel foods, are also met.
By December 2022, the European Commission had received about 190 novel food applications for food products containing CBD, either produced synthetically or extracted from Cannabis sativa. The intended use mainly relates to ingredients for food supplements rather than ingredients in other foods. Many of these applications are incomplete, with poor data in parts, while a common procedural error made in some of these applications has been to simply rely on safety results of CBD submitted for the medicinal product Epidyolex, which now has EU-wide marketing approval. As the data related to the safety results of Epidyolex is the intellectual property of that applicant it cannot be transferred to another applicant without permission from the owner of the intellectual property rights.
When starting to evaluate some of the CBD applications, it became clear to EFSA that there are knowledge gaps that need to be addressed before a conclusion on the safety of CBD can be reached, including its possible effects on the liver, gastrointestinal tract, endocrine system, nervous system and psychological function, as well as concerns regarding reproductive toxicity. There are data gaps in the research regarding drug-drug interactions of CBD and the effects of increasing solubility for bioavailability (the proportion of a substance which enters the circulation when introduced into the body to have an active effect), both of which can have the effect of making substances more potent than originally expected. ‘Full spectrum’ product applications that contain whole cannabis plant extracts are even more complex as they comprise a mixture of compounds. Finally, side effects for novel foods are not acceptable as compared to medicines, which may allow some undesirable side effects in order to prevent or treat a disease when the benefits are judged to be greater than the risks. As there have been a large number of incoming applications for CBD-containing food products, EFSA adopted in May 2022 a statement on the safety of CBD as a novel food. Considering the significant uncertainties and data gaps outlined above, the EFSA panel on nutrition, novel foods and food allergens (NDA) concluded that at this date the safety of CBD as a novel food, that is, a product with a safe daily intake for humans, cannot be established.
Is industrial cannabis legal?
In the European Union it is legal to cultivate and supply cannabis plants for hemp fibre and seeds if they have low levels of THC. Rules on the marketing of seeds of oil and fibre plants are laid down in Council Directive 2002/57/EC. Only varieties of hemp listed in the European Union Common catalogue of varieties of agricultural plant species (the ‘Plant variety database’) can be marketed in the European Union for agricultural purposes, and some varieties may be cultivated and supplied for hemp fibre. The relevant EU regulations are listed below (4).
When cultivating hemp, farmers are only eligible for support under the common agricultural policy when using certified seed of specified hemp varieties; only varieties with a THC content not exceeding 0.3 % may be used (Regulation (EU) No 2021/2115). Standardised procedures for the determination of their THC content state that, for example, the samples to be tested for THC should be taken from the top 30 centimetres of the plant, with at least one female flower, dried, with stems and seeds removed (Commission Delegated Regulation (EU) No 639/2014, Annex III).
According to the Court of Justice of the European Union (CJEU), case C-207/08 (Babanov), the cultivation of hemp fulfilling the strict conditions in the EU legislation cannot be prohibited in any Member State, as this would be in conflict with EU law. New countries joining the European Union whose national narcotic control laws may have stipulated that it is illegal to grow any cannabis plant have sometimes needed to change their laws in order to permit this exception. This case provides an example of where differences in national drug control approaches can be incompatible with EU regulations.
The EU Common catalogue of varieties of agricultural plant species lists varieties whose seed can be marketed throughout the European Union, including approximately 100 varieties of the species ‘hemp: Cannabis sativa’ that have been notified by 14 Member States. The number has increased by 30 varieties since 2019 (European Commission, 2022). Only certified seeds of agricultural cannabis varieties may be marketed in the European Union, in line with the Council Directive 2002/57/EC on the marketing of seed of oil and fibre plants. The seed certification carried out in Member States ensures the identity, health and quality of seeds for farmers. Some EU Member States may explicitly exclude all derivatives of those varieties from their narcotic schedules. This includes all parts of the plant, seeds, extracts and tinctures, as well as the resin. An amendment to Regulation (EC) No 1881/2006 (Commission Regulation (EU) 2022/1393) establishes the maximum levels of delta-9-THC (sum of delta-9-THC and delta-9-THCA, expressed as delta-9-THC) in hemp seeds of 3.0 milligrams per kilogram, in hemp seed oil of 7.5 milligrams per kilogram and for other hemp seed derived products of 3 milligrams per kilogram.
Imports of raw hemp and hemp seeds are also subject to certain conditions to ensure that the maximum THC limit is respected (Regulation (EU) No 1308/2013). It should be stressed, however, that this legislative framework was developed for the hemp industry (raw hemp and seeds) and was not intended to have implications for other products or product safety in humans. Importantly, the application of a safety limit expressed as a percentage (e.g. 0.2 % or 0.3 % THC limit) is not appropriate for products intended for human consumption as the actual dose ingested will be dependent on the volume of the product consumed. Additionally, intra-EU trade in hemp products may remain subject to national legislation.
Is it possible to trademark cannabis varieties or cannabis products?
It is possible to protect the intellectual property rights of plant varieties, seeds and products, which require distinct applications and authorisations. The EU Community Plant Variety Office (CPVO) is responsible for issuing intellectual property rights (Community plant variety rights, CPVR) for a new cannabis variety if it fulfils certain criteria. The requirements for protecting a plant variety are novelty, distinctness, uniformity, stability (the ‘DUS criteria’) and a suitable denomination. The intended use (fibre, oil, chemicals, etc.) and the content of THC must be specified in the application to ensure correct classification of the plant variety.
Varieties yielding any content of THC may still be registered nationally or at the European level to protect plant breeders’ rights, but to be marketed they have to fulfil the requirements for trading.
While there were approximately 22 applications to register new varieties of cannabis over the period 1999–2015, with most of them being seed-propagated varieties mainly used for fibre, woody core or oil seed, this increased to approximately 300 applications between 2018 and 2021, mostly originating from plant breeders in the Netherlands, Spain and Italy. Over 80 % of these applications are for ‘other than fibre use, vegetatively propagated’ (see Figure 9).
In recent years the European Union Intellectual Property Office (EUIPO) has also seen a considerable increase in applications to register trademarks and designs containing references to cannabis. Its Guidelines for Examination (5) have clarified that applications may be refused as being contrary to public policy or acceptable principles of morality (Article 7(1)(f) EU trade mark Regulation 2017/1001) if they could be perceived as promoting the illegal use of drugs. As national legislations generally prohibit drug use and aim to counter drug supply, this represents an objective basis for refusal if the product is not intended for medicinal use.
The exponential increase in the licit use of cannabis plants or products and related applications for authorisation has highlighted a number of challenges; for example, the complexity of procedures to transport cannabis-related materials and products, such as propagated plant material or THC products, to CPVO offices for testing. Moreover, individual laboratories in different countries might not use harmonised or standardised techniques, which could complicate obtaining consistent results when assessing THC levels in different forms of cannabis (powders, oil, edibles, herbal material, etc.). Obtaining differing results may further complicate authorisations for transport, and some customs authorities are currently working on a solution for this issue.

Source: Community Plant Variety Office.
Show a text version of the above graphic
Flow chart for the classification of new cannabis varieties into types
THC content lower than 0.2 %
-
If the THC content is lower than 0.2 %:
- Check if it is intended for the production of oil seed or fibre.
- If YES:
- Method of propagation:
- Seed (excluding feminised seed): Type A
- Vegetatively propagated: Type B
- Feminised seed: Type E
- Method of propagation:
- If NO:
- Method of propagation:
- Vegetatively propagated: Type C
- Feminised seed: Type D
- Method of propagation:
- If YES:
- Check if it is intended for the production of oil seed or fibre.
-
If the content is higher than 0.2 %:
- Method of propagation:
- Vegetatively propagated: Type C
- Feminised seed: Type D
- Method of propagation:
#EMBED-cannabis-laws-faq-box-11#
Find out more
- Low-THC cannabis products in Europe, EMCDDA, 2020.
- THC sampling procedure: Commission Delegated Regulation (EU) No 639/2014.
- Tobacco Products Directive 2014/40/EU.
- European Commission, EU cosmetics regulations.
- Food safety in the European Union, European Union.
- Scientific report on ‘Acute human exposure assessment to THC’, EFSA, November 2019. Statement on cannabidiol as a novel food: data gaps and uncertainties, EFSA Journal, June 2022.
- EU Plant variety database.
- Medical use of cannabis and cannabinoids: questions and answers for policymaking, EMCDDA, 2018.
- A summary of reviews of evidence on the efficacy and safety of medical use of cannabis and cannabinoids, background paper by Wayne Hall, 2018.
- For further information on the EU regulatory framework see:
- European Medicines Agency, About us, What we do.
- European Commission, Authorisation procedures: The centralised procedure.
- WHO Expert Committee on Drug Dependence, Critical review of cannabis and associated substances.
- EU Community Plant Variety Office.
- EU Intellectual Property Office.
(1) Hall, 2018, provides a general description of the available evidence on the efficacy and safety of cannabis/cannabinoids. It is important to note that there are differences between this and the evidence that is required to apply for the authorisation of a particular medicine with a specific active substance and presented for a specific therapeutic indication, for which product-specific quality, non-clinical and clinical data will have to be generated and submitted for assessment.
(2) This is not an exhaustive overview of all potentially relevant EU regulations that may apply to cannabis as cosmetic products.
(3) This is not an exhaustive overview of all potentially relevant EU regulations that may apply to cannabis as food products.
(4) This is not an exhaustive overview of all potentially relevant EU regulations that may apply to cannabis as industrial products.
(5) EUIPO, Trade mark and designs guidelines, Chapter 7 Trade marks contrary to public policy or acceptable principles of morality (Article 7 (1)(f) EUTMR), 3. Accepted Principles of Morality, Edition 2022.
Text boxes
Available text boxes:
- Examples of cannabis-derived medicinal products in Europe
- Example: Cannabis-related trademark applications








